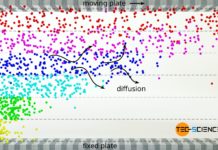
Viscosity of an ideal gas
The viscosity of ideal gases is mainly based on the momentum transfer due to diffusion between the fluid layers.
Definition of viscosity
In the article Viscosity, the cause of viscosity was mainly attributed...
Internal energy & heat capacity of ideal gases (kinetic theory of gases)
In this article, learn more about the relationship between internal energy and heat capacity in connection with the kinetic theory of gases.
https://www.youtube.com/watch?v=M9i8YfBu30w
Internal energy
In the article equipartition theorem it has already been...
Determination of the speed distribution in a gas
Learn more about experimentally determining the velocity distribution of molecules in gases in this article.
https://www.youtube.com/watch?v=OVcen-FWc-4
Introduction
As already explained in the article Temperature and particle motion, the temperature of a gas is a...
Pressure and temperature (kinetic theory of gases)
In this article, learn more about the relationship between pressure and temperature in connection with the kinetic theory of gases.
https://www.youtube.com/watch?v=3NYFD9LMV2A
Introduction
In order to connect the macroscopically observed state variables of a gas...
Equipartition theorem
The equipartition theorem states that the kinetic energy of the gas molecules is equally divided along all three spatial directions!
https://www.youtube.com/watch?v=wxFbzgYe9cM
Equipartition theorem
In the article Pressure and temperature the following equation was derived...
Why do liquids evaporate?
In this article, learn how the evaporation of liquids can be qualitatively explained using the Maxwell-Boltzmann distribution.
Maxwell-Boltzmann distribution of ideal gases
The figure below shows the speed distribution according to Maxwell-Boltzmann for...