In this article we will deal with the design and operation of a thermocouple which is used to measure temperatures.
Seebeck effect
A thermocouple uses the phenomenon that a voltage is caused by the presence of a temperature gradient along an electrical conductor.

The cause of this thermoelectric effect lies in the metallic bonding of metals and in particular in the free electrons. If a metal wire is only heated at one end, the lattice oscillations and the movements of the free electrons increase there. Because of the heavy collisions they begin to spread and diffuse to the cold end. There, the kinetic energies of the electrons are lower and the electrons are not repelled again by heavy collisions.
The electron gas behaves literally like a “real gas” whose particles occupy a larger space when heated and the particle density decreases accordingly.
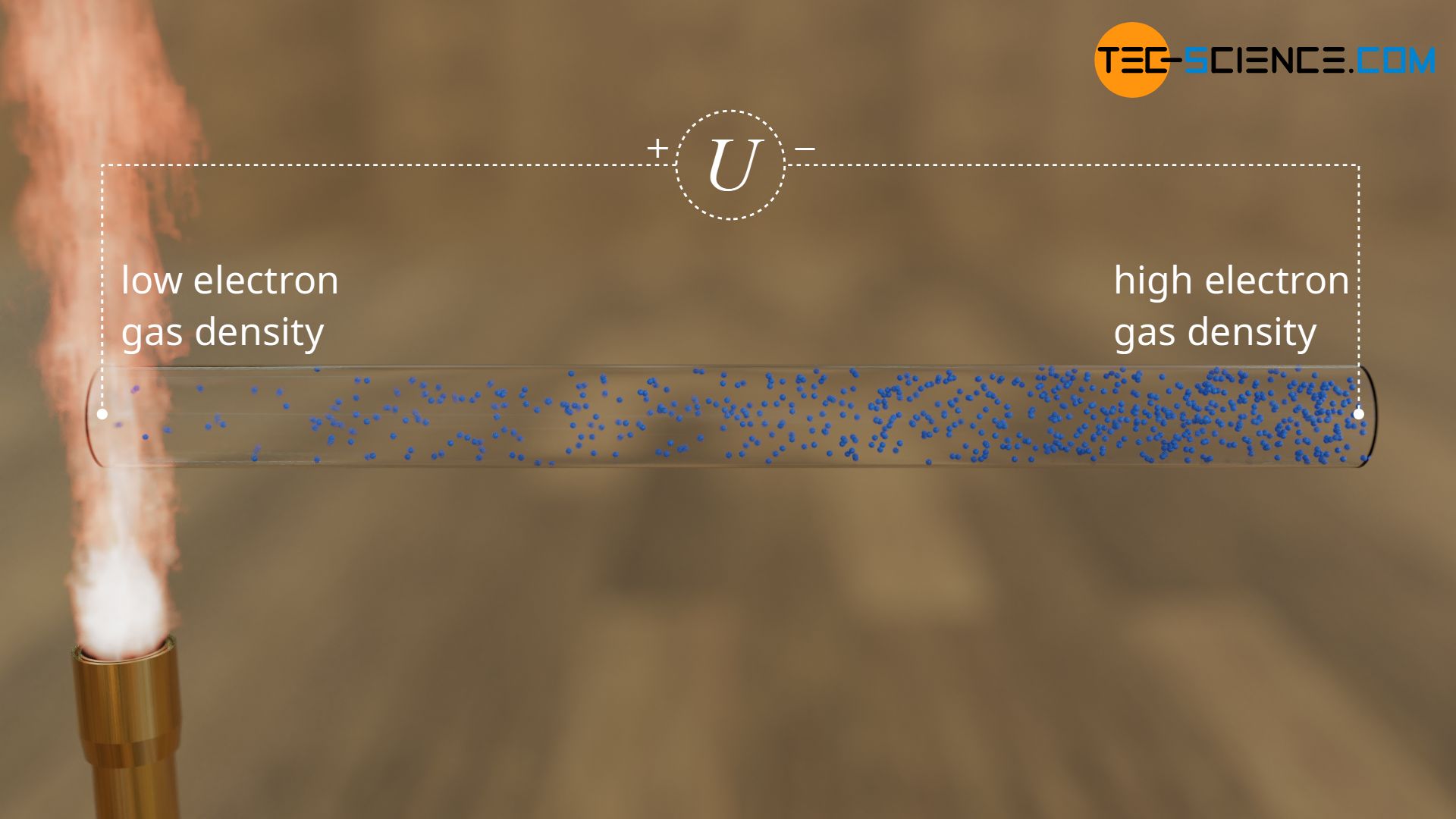
The hot end of the wire thus has a smaller number of electrons than the cold end. As a result, an electrical voltage is obtained between the two ends, also known as thermoelectric voltage. This thermoelectric effect is also called the Seebeck effect and the thermoelectric voltage is also referred to as Seebeck voltage.
The thermoelectric effect (Seebeck effect) is a phenomenon in which heat is converted into electrical energy by diffusion of charged particles due to temperature gradients!

Per Kelvin temperature difference along one electrical conductor, the Seebeck effect is in the order of a few microvolts. The thermoelectric voltage is strongly dependent on the conductor material at a given temperature gradient. In the case of materials whose free electrons can be set in motion relatively easily when heated, the thermoelectric voltage is greater because the electrons are more strongly spread.
Thermocouple
If a simple wire such as the one described above were exposed to a heat source, the wire would heat up evenly. Due to the lack of temperature gradient between the ends, no thermoelectric voltage could be measured. In addition, further thermoelectric voltages would be caused at the additional measuring leads.
Therefore, two different conductors are required which differ in the strength of the Seebeck effect (e.g. copper and iron). The different wires are now connected at one end. This joint serves as a measuring junction (“hot junction”) and is exposed to the temperature to be measured. The other ends lead to the so-called reference junction (“cold junction”), the temperature of which is usually ambient temperature.
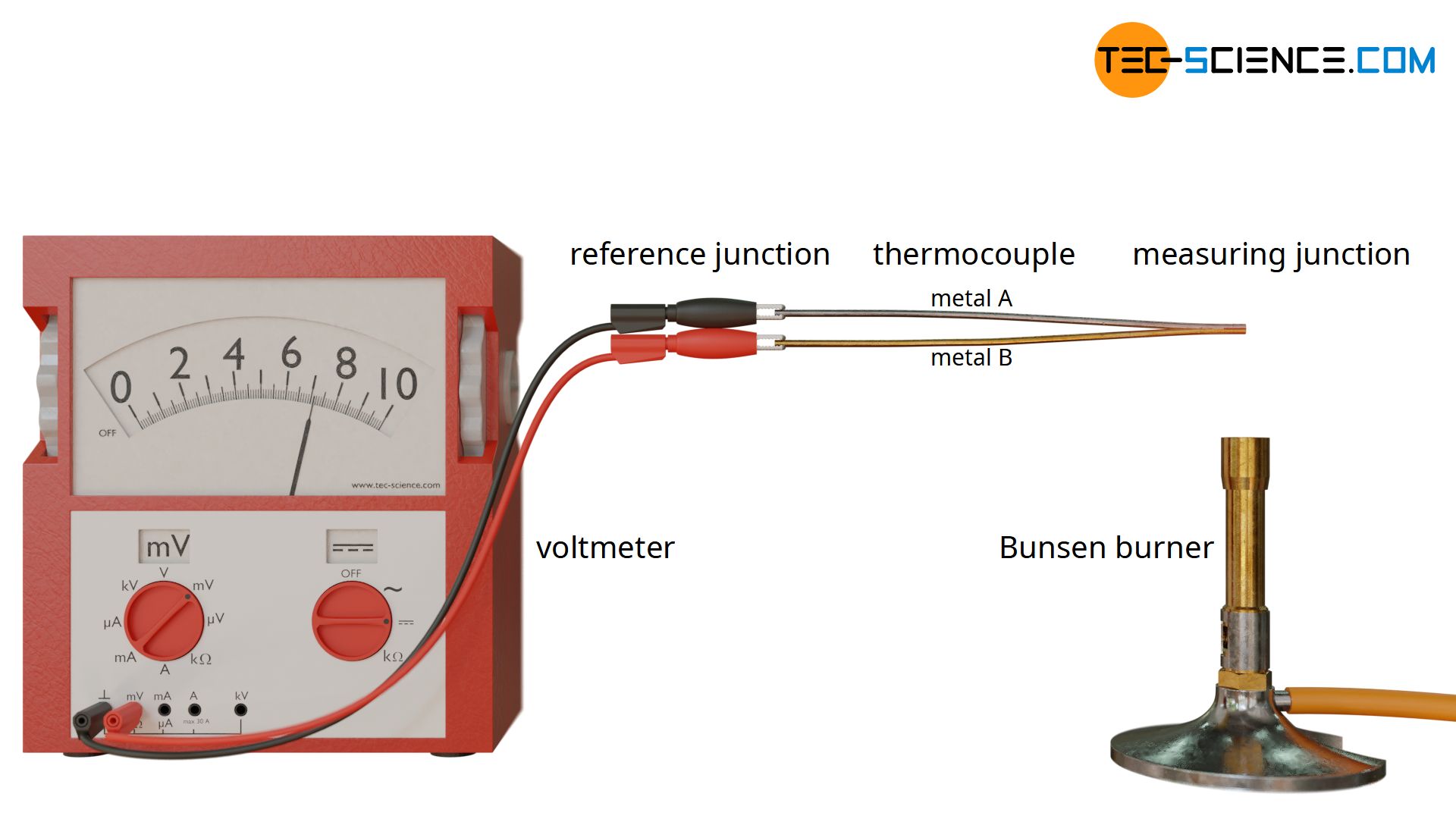
There is now a temperature gradient between the measuring junction and the reference junction and thus between the ends of the respective wires. This results in a thermoelectric effect with the consequence of an electrical voltage. Since these are different metals, the thermoelectric effect is different in strength. For example, compared to copper, iron has a thermoelectric voltage around 6 times as high.
Since the two metals are connected to each other at the measuring junction, they are at the same electric potential. As a result, the electric potentials at the reference junction differ and an electrical voltage is caused that can be measured with a voltmeter.
This electrical voltage can also be explained as follows: The diffusion of electrons from the measuring point to the reference point due to the temperature gradient is much more intense with iron, i.e. more electrons will accumulate there than at the end of the copper wire. This leads to a charge imbalance between the wire ends at the reference junction, in favour of increased electron accumulation at the end of the iron wire. A measurable voltage is generated at the reference junction, which is dependent on the temperature difference to the measuring junction.
Thermocouples only measure thermoelectric voltages due to temperature differences between measuring junction and reference junction!

Cold junction compensation
With the measurement setup described above, the measured voltage can initially only be used to determine the temperature difference between the measuring junction and the reference junction. To determine the absolute temperature at the measuring point, the temperature at the reference junction must therefore be known. This determination of the temperature at the “cold” junction is also referred to as cold junction compensation.
The cold junction temperature can, for example, be determined with a Pt100, whose electrical signal can then be processed directly with the thermoelectric voltage in order to output the temperature at the measuring point.
Types of thermocouples
The selection of materials to generate the thermoelectric voltage of a thermocouple is of central importance. The following properties of the material combination are taken as a basis:
- highest possible thermoelectric voltages
- highest possible corrosion resistance
- linear characteristic between temperature difference and thermoelectric voltage (high accuracy)
- linearity over a wide temperature range
Up to now, no material pairing has been able to cover all required properties to the same extent. Therefore, different material combinations (so-called types) are used depending on the application, e.g.:
- Type J: copper-nickel alloy & iron; can be used from approx. -200 °C to 750 °C
- Type K: nickel-chromium alloy & nickel; can be used from approx. -250 °C to 1200 °C
- Type R: platinum-rhodium alloy & platinum; can be used from approx. -50°C to 1500 °C
Type K is the most common type in private use.
Pros
The advantage of thermocouples is their relatively short delay time, i.e. they react very quickly to changes in temperature. In addition, thermocouples are mechanically very robust and cover a temperature range from -250 °C to +2000 °C depending on the type. In addition, due to their space-saving design, thermocouples can also easily reach hard-to-reach locations. Due to the relatively small measuring tip, thermocouples can also measure temperatures of very small objects.